Hort 550 Lab
Protein: Arabidopsis thaliana DAHP Synthase 1
Gene: At4G39980
Entryway Enzyme of the Shikimate Pathway
At4G39980 is the gene which encodes AtDHS1, the first of three isozymes of 3-deoxy-D-arabinoheptulosonate-7-phosphate (DAHP) synthase in Arabidopsis thaliana (arabidopsis.org). DAHP synthase is the entryway enzyme of the shikimate pathway; this biosynthetic pathway leads to a diverse set of aromatic compounds (Maeda and Dudareva, 2012). The major products are lignin, a cell wall component, and the three aromatic amino acids; these combined can take up to 30% of a plant's carbon (Maeda and Dudareva, 2012). As a plant must synthesize more cell wall material and more protein to grow, it is logical that at least one isozyme of DAHP synthase would be correlated to growth; I believe AtDHS1 is so correlated.
I am presently engaged in elucidating the nature of the redox regulation of AtDHS1 under the supervision of Professor Hiroshi Maeda of the Botany department. It is known that AtDHS1 and many other plant DAHP synthases are redox-regulated, requiring electrons from thioredoxin to function (Entus et al, 2002). It is also known that the DAHP synthase of Mycobacterium tuberculosis is redox-regulated; structural studies have pinpointed two cysteine residues, Cys87 and Cys440, which are likely responsible (Webby et al, 2005). Cys87 binds the essential metal (usually manganese) ion; it is thought that Cys440 can rotate, form a disulfide bond with Cys87, and prevent it from binding that metal ion. A reducing agent can then add electrons to break said disulfide bond, allowing Cys87 to once again bind the metal ion. These two cysteines are conserved in Arabidopsis thaliana DAHP synthases; the primary sequence aligns well, and a hypothetical structure generated via SWISS-MODEL fits as well.
The thioredoxin necessary to activate the enzyme is found primarily during the day. Daylight, via photosynthesis, provides energy and reducing power for biosynthesis; as such, it is only sensible that the enzyme would be activated during the day (by provision of thioredoxin) and inactivated by night.
I am presently engaged in elucidating the nature of the redox regulation of AtDHS1 under the supervision of Professor Hiroshi Maeda of the Botany department. It is known that AtDHS1 and many other plant DAHP synthases are redox-regulated, requiring electrons from thioredoxin to function (Entus et al, 2002). It is also known that the DAHP synthase of Mycobacterium tuberculosis is redox-regulated; structural studies have pinpointed two cysteine residues, Cys87 and Cys440, which are likely responsible (Webby et al, 2005). Cys87 binds the essential metal (usually manganese) ion; it is thought that Cys440 can rotate, form a disulfide bond with Cys87, and prevent it from binding that metal ion. A reducing agent can then add electrons to break said disulfide bond, allowing Cys87 to once again bind the metal ion. These two cysteines are conserved in Arabidopsis thaliana DAHP synthases; the primary sequence aligns well, and a hypothetical structure generated via SWISS-MODEL fits as well.
The thioredoxin necessary to activate the enzyme is found primarily during the day. Daylight, via photosynthesis, provides energy and reducing power for biosynthesis; as such, it is only sensible that the enzyme would be activated during the day (by provision of thioredoxin) and inactivated by night.
Shikimate Pathway

DAHP synthase is the entryway enzyme of the shikimate pathway, responsible for synthesizing a large number of aromatic compounds. Image is adapted from Maeda and Dudareva, 2012.
Primary Alignment of Type II DAHP Synthases

Residues in black are perfectly conserved through all enzymes. The white residue is not conserved. Highlighted in red are the cysteine residues implicated in redox regulation of DAHP synthase.
Hypothetical Structure of AtDHS1

A model of hypothetical AtDHS1 structure against 3NV8, the type II DAHP synthase of M. tuberculosis. Numeric labels correspond to Cys139 and Cys494 of AtDHS1; the unlabeled residues are Cys87 and Cys440 of 3NV8. The purple sphere is a manganese ion, considered essential for protein function (Bordoli et al, 2006. Kiefer et al, 2009. Peitsch, 1995. PyMol). Notice how it would be easy for Cys494 to rotate up and form a disulfide bond with Cys139.
DAHP Synthase: Aldol Condensation of PEP and E4P
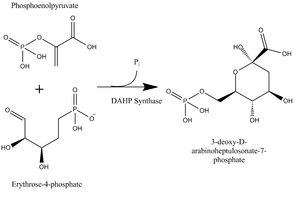
DAHP synthase catalyzes an aldol condensation of phosophoenolpyruvate (PEP) and erythrose-4-phosphate (E4P) into a single molecule of 3-deoxy-D-arabinoheptulosonate-7-phosphate (DAHP). PEP is a glycolysis intermediate, and E4P a pentose phosphate pathway intermediate, thus both relatively common in the cell (Nelson and Cox, 2008). DAHP is the first committed intermediate of the shikimate pathway (Maeda and Dudareva, 2012).
Redox Regulation: Disulfide Bonds
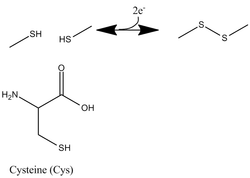
Cysteine residues are notable for their ability to oxidize and form disulfide bonds. Reducing power, often in the form of thioredoxin (in vivo) or dithiothreitol (in vitro), however, can break the disulfide bond, freeing the cysteines' sulfur atoms to interact elsewhere.
Plastid Import Sequence
The ChloroP tool takes a primary sequence, and predicts any plastid import sequences. Analysis of AtDHS1 showed a predicted sequence of 47 amino acids.
### chlorop v1.1 prediction results* ###########################
*Note: Truncated to show only AtDHS1
Number of query sequences: 8
Name Length Score cTP CS- cTP-
score length
---------------------------------------------------------------
gi_20141225_sp_P2997 525 0.578 Y 5.927 47
---------------------------------------------------------------
### chlorop v1.1 prediction results* ###########################
*Note: Truncated to show only AtDHS1
Number of query sequences: 8
Name Length Score cTP CS- cTP-
score length
---------------------------------------------------------------
gi_20141225_sp_P2997 525 0.578 Y 5.927 47
---------------------------------------------------------------
